Acute myeloid leukemia with complex karyotype (CK-AML), the second largest cytogenetic subset in AML (1), commonly (70-80%) has TP53 mutations (2) and is associate with a dismal outcome for patients treated with current therapeutic options (3). Multiple attempts to target a specific mutation in CK-AML have failed, mainly because when a particular mutation is inhibited, the leukemic cells may survive and proliferate through other genetic aberrations which they harbor (4).
The cellular glutamine state is a promising target to selectively eradicate CK-AML cells irrespective of their genetic signature, because it is a fundamental property of each cell that differs sufficiently between leukemic and normal cells (5). Growing evidence suggests that AML cells, compared to normal cells, have an increased susceptibility to the disruption of glutamine metabolism (6). Asparaginase converts asparagine and glutamine to aspartate and glutamate, respectively, decreasing plasma concentrations of asparagine and glutamine (7). Asparaginase products have been reported to induce rapid and complete depletion of intracellular glutamine in AML cell lines and primary AML samples regardless of their mutational status (5, 6, 8) and the anti-leukemic activity of asparaginases have been shown to correlate with their glutaminase activity levels(6).Long-acting crisantaspase, Pegcrisantaspase (PegC), is a recombinant pegylated asparaginase that has been used in pediatric patients with ALL (9). Interference with glutamine metabolism has been shown to overcome resistance to BCL-2 inhibition in AML and other cell types (10-12). Venetoclax (Ven), a BCL-2 specific inhibitor has demonstrated anticancer efficacy, and is indicated in combination with azacitidine, decitabine or low-dose cytarabine to treat newly-diagnosed AML in adults age 75 years or older. We therefore hypothesized that depletion of glutamine induced by PegC would not only inhibit proliferation of CK-AML but also enhance the apoptotic activity of Ven-mediated antagonism of BCL-2 in CK-AML.
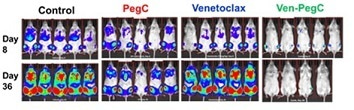
In our study published in Leukemia (PMID: 33199836), we report potent anti-leukemic activity of the Ven-PegC combination in CK-AML cell lines, primary cells, and in a patient-derived xenograft mouse model. We demonstrated that Ven-PegC blocks synthesis of proteins in CK-AML cells by enhancing eIF4E-4EBP1 interaction on the cap-binding complex thereby inhibiting cap-dependent translation of mRNA. In vivo, Ven-PegC treatment exhibited remarkable efficacy and was able to completely deplete plasma glutamine and asparagine. Although BCL-2 inhibition with Ven is now commonly used for AML patients, our proposed combination of Ven and PegC is a novel, mechanistically-based regimen that is unique to the field and has the advantage of combining two drugs already in clinical use for acute leukemias. These promising pre-clinical data have prompted us to design a Phase 1 clinical trial of Ven-PegC for treatment of adult subjects with R/R AML including AML with complex karyotype and/or TP53 mutation.

Ven-PegC impedes cap-dependent translation and protein synthesis.
- M. L. Slovak et al., Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: a Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood96, 4075-4083 (2000).
- C. Schoch et al., Acute myeloid leukemia with a complex aberrant karyotype is a distinct biological entity characterized by genomic imbalances and a specific gene expression profile. Genes Chromosomes Cancer43, 227-238 (2005).
- S. O. Ciurea et al., Relapse and survival after transplantation for complex karyotype acute myeloid leukemia: A report from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation and the University of Texas MD Anderson Cancer Center. Cancer124, 2134-2141 (2018).
- J. P. Patel et al., Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N Engl J Med366, 1079-1089 (2012).
- M. Goto et al., Importance of glutamine metabolism in leukemia cells by energy production through TCA cycle and by redox homeostasis. Cancer Invest32, 241-247 (2014).
- L. Willems et al., Inhibiting glutamine uptake represents an attractive new strategy for treating acute myeloid leukemia. Blood122, 3521-3532 (2013).
- A. Beckett, D. Gervais, What makes a good new therapeutic L-asparaginase? World J Microbiol Biotechnol35, 152 (2019).
- I. Samudio, M. Konopleva, Asparaginase unveils glutamine-addicted AML. Blood122, 3398-3400 (2013).
- R. E. Rau et al., Outcome of pediatric patients with acute lymphoblastic leukemia/lymphoblastic lymphoma with hypersensitivity to pegaspargase treated with PEGylated Erwinia asparaginase, pegcrisantaspase: A report from the Children's Oncology Group. Pediatric blood & cancer65, (2018).
- N. Jacque et al., Targeting glutaminolysis has antileukemic activity in acute myeloid leukemia and synergizes with BCL-2 inhibition. Blood126, 1346-1356 (2015).
- R. Bajpai et al., Targeting glutamine metabolism in multiple myeloma enhances BIM binding to BCL-2 eliciting synthetic lethality to venetoclax. Oncogene35, 3955-3964 (2016).
- W. K. Chang, K. D. Yang, H. Chuang, J. T. Jan, M. F. Shaio, Glutamine protects activated human T cells from apoptosis by up-regulating glutathione and Bcl-2 levels. Clin Immunol104, 151-160 (2002).






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in