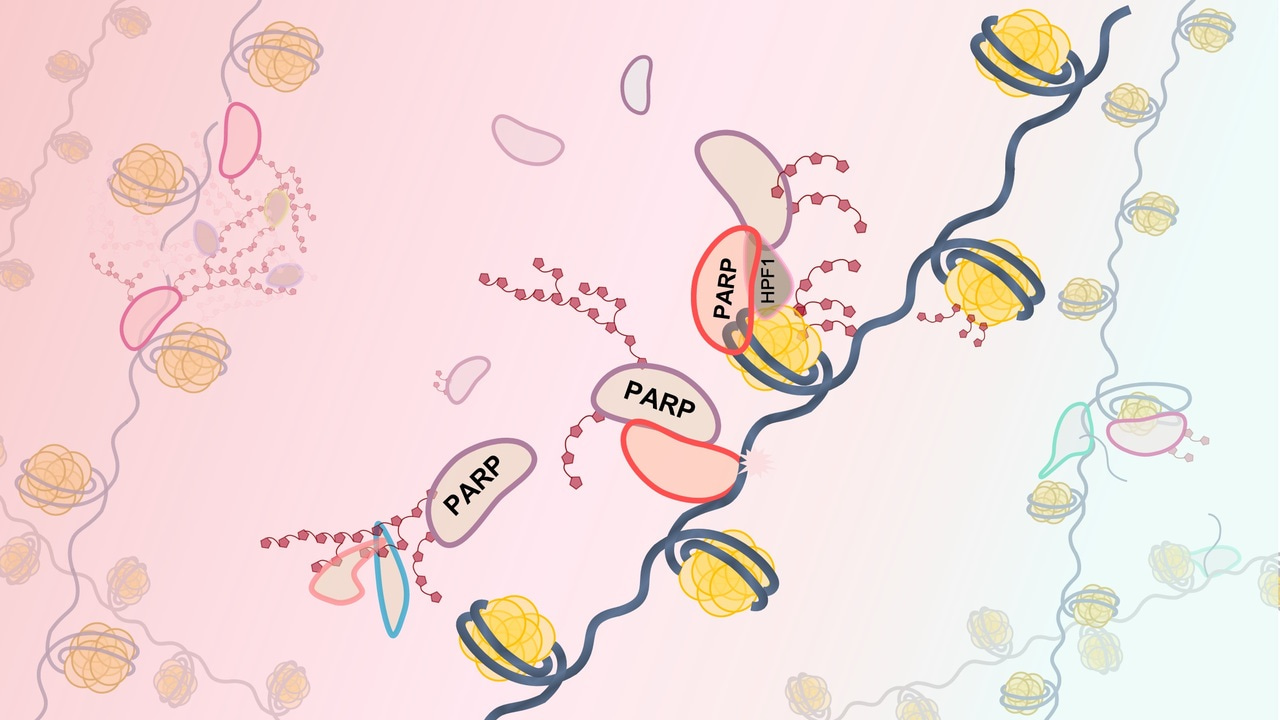
Just imagine: well-known enzyme, which regulates key cell processes responsible for genome stability suddenly, acquires a cofactor that radically changes its specificity. About 2000 articles since 1963 with come from tremendous effort and many hours of work. And, suddenly, it turns out that this enzyme can form a joint active site with a recently discovered protein factor. This factor changes the specificity of the reaction catalyzed with enzyme, it changes the reaction rate, and it seems that it is necessary to study everything (or almost everything) from scratch. This is about PARP1/2 enzymes, and their new cofactor HPF1.
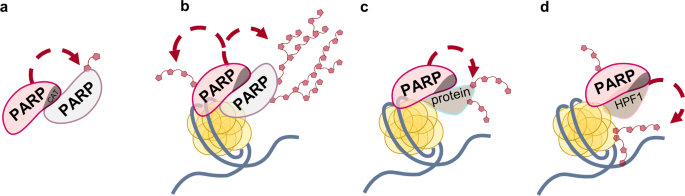
PARPs synthesize poly(ADP-ribose) (PAR) composed of linear and/or branched repeats of ADP-ribose. PARylation is a dynamic post-translational modification of biomolecules that plays an indispensable role in regulating of a number of biological processes, including DNA damage response. HPF1 was shown to complete the active site of PARPs via complex formation, and the role of the joint active site was suggested to switch the PARylation specificity to serine residues. HPF1 plays an essential role in the PARP1- and PARP2-catalyzed PARylation of histones. How is HPF1 involved in the response to DNA damage? Is all PAR synthesis in the cell HPF1-dependent? How do PARP inhibitors, known anti-cancer drugs, affect HPF1-dependent PARylation? How does histone PARylation affect chromatin structure? Now we have a lot of questions. And here, we do a little step for mankind, but giant leap for us: we show dual influence of HPF1 on PARPs activity using classical biochemical methods (as our teachers bequeathed us). In the present study, we compared HPF1 effects on the PARP1 and PARP2 activities in the autoPARylation reaction performed in the presence of damaged DNA or nucleosome, as well as in the heteroPARylation of histones. Recently, the capability of HPF1 to limit the level of PARP1 automodification via shortening of PAR chain was shown. It can be explained by the interaction of HPF1 with His residue (His826 and His381 in PARP1 and PARP2, respectively), which is critical for PAR chain elongation. In our study, HPF1 was found to stimulate the automodification of both PARP1 and PARP2 in a concentration-dependent manner, with the effects detected for PARP2 exceeding those for PARP1. PARP2 was revealed to catalyze more efficiently modification of histones than the automodification.
Data obtained previously and here under different experimental conditions provide evidence that HPF1 can promote opposite effects on distinct stages of the PARP1- and PARP2-catalyzed reaction: it stimulates early stages and inhibits elongation by shielding of amino acid residues important for PAR chain elongation. Thus, HPF1 can produce different effects on the overall PAR synthesis at low and high NAD+ concentrations. Probably, at low NAD+ concentrations, the initiation has the greatest contribution to the total reaction output, and the HPF1-induced stimulation of early stages should significantly increase the level of protein-bound ADP-ribose. In contrast, at high NAD+ concentrations, the elongation has a predominant contribution to PAR synthesis. The inhibitory action of HPF1 on the elongation step may therefore suppress the PARylation at higher NAD+ concentrations. Thus, the hyperactivation of PARP in PAR synthesis at high NAD+ concentrations is inhibited by HPF1. At low NAD+ concentrations, the activity of PARP is stimulated by HPF1, with the optimal concentration and extent of stimulation being dependent on the relative amounts of DNA-free and DNA-bound PARP.

The dual regulatory function of HPF1 may contribute to maintenance of PARP activity at the level required for its function in the DNA damage response signaling and repair, independently on the NAD+ concentrations. The suppression is most likely predominant function of HPF1 at the normal NAD+ concentration in the nucleus (100 μM) to prevent negative consequences of PARP hyperactivation. The stimulatory action of HPF1 might regulate PARP activity under stress conditions, when the sustained PARP activity leads to NAD+ depletion.
If you are intrigued, or if you come across too many unfamiliar words here, we recommend that you read our original manuscript [Kurgina TA, Moor NA, Kutuzov MM, Naumenko KN, Ukraintsev AA, Lavrik OI. Dual function of HPF1 in the modulation of PARP1 and PARP2 activities. Commun Biol. 2021 Nov 3;4(1):1259. doi: 10.1038/s42003-021-02780-0. PMID: 34732825] and figure it out. Welcome!





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in