Since the 80’s, the Laboratory of Immunology and Biology of Metastasis aimed to understand the mechanisms of metastatic spread of cancer cells, in particular of HER2 positive breast cancer and musculoskeletal sarcomas, and to the definition of systemic strategies directed against specific molecular targets, based on pharmacological and immunological agents. The laboratory team developed a long and consolidated experience in the development of preclinical models useful for studies on tumor-host interactions. In this context, our preclinical model of HER2 dynamic expression was born.

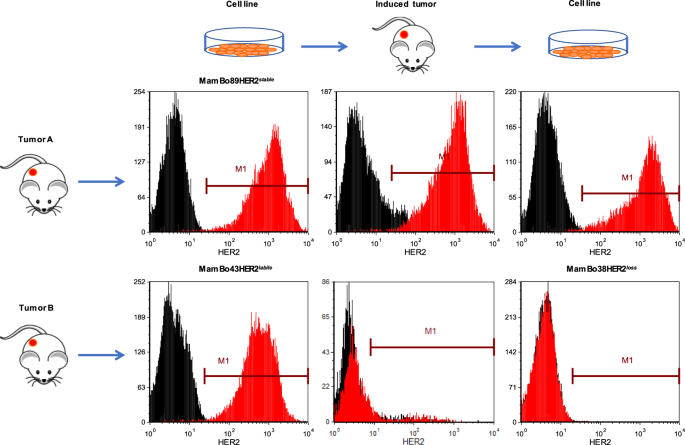
MamBo89HER2stable and MamBo43HER2labile are two HER2-positive cell lines derived from two mammary tumors of human HER2 transgenic mice, showing different dynamics of HER2 expression. MamBo89HER2stable cell line displayed high and stable HER2 expression, both in vitro and in vivo, whereas MamBo43HER2labile undergo HER2 loss upon in vitro treatment with trastuzumab or in vivo tumor growth, the HER2-negative cell line MamBo38HER2loss was derived from one of such tumor. MamBo38HER2loss cells showed a epithelial to mesenchymal transition, high stemness and high in vivo malignancy.

This dynamics of HER2 expression may mirror receptor conversion during human tumor progression patient progressed tumors. In patients, HER2 loss occurred in more than 20% of cases with a HER2-positive primary tumor and has been associated with a worse survival rate (Schrijver W et al., Receptor conversion in distant breast cancer metastases: a systematic review and meta-analysis. J Natl Cancer Inst. 2018).

Two questions then popped up in our minds: What caused the loss of HER2 expression? What might be a therapeutic target in cells that have lost expression of the HER2 oncogene?
We don’t know yet why HER2 expression is lost in our model. The HER2 copy number of MamBo38HER2loss cells was similar to the HER2-positive MamBo43HER2labile cells, ruling out a total loss of the transgene. However, the level of HER2 transcript was barely detectable in MamBo38HER2loss cells as compared to MamBo89HER2stable and MamBo43HER2labile cells and western blot analysis showed a complete lack of HER2 proteins within the HER2-negative cells. To determine whether DNA methylation could be involved in the loss of HER2 expression, MamBo38HER2loss cells were treated with 5-Aza-2’-deoxycytidine, a DNA Methil-transferase inhibitor, but HER2 expression was not induced. We are now studying whether other epigenetic modulators could be effective in unblocking HER2 expression in MamBo38HER2loss cell line.
Although understanding the mechanism behind HER2 loss was becoming increasingly difficult, we decide to not lose heart and keep looking for a valid therapeutic target to stop the growth of HER2 negative cell line MamBo38HER2loss.
The comparison between differentially expressed genes in HER2-negative and HER2-positive cell lines shed light on the biological processes involved in epithelial-to-mesenchymal transition (EMT). The high expression of EMT-associated genes and the stemness profile suggested us that we have in our hands (…sorry flasks!) cells resembling claudin-low human tumors. Thus, also in patients HER2-positive primary lesions can progress to claudin-lowness.
With the help of RNA-sequencing analysis we identified platelet-derived growth factor receptor beta (PDGFR-β) as a possible pathway that sustains the malignant phenotype of MamBo38HER2loss cell line that have lost their addiction to HER2. PDGFR-β is known to be involved in EMT promotion, stemness phenotype, it is a pericyte marker and is a druggable target by sunitinib.

Sunitinib is a multi-targeted molecule that offers the possibility of inhibiting VEGFR, which was also identified as being up-regulated in HER2-negative cells in our analysis. Sunitinib was effective in impairing (not eradicating) the growth of MamBo38HER2loss cells and the emergence of HER2-negative tumors from MamBo43HER2labile cells. These data pave suggest that the use of sunitinib or similar drugs might be useful in the treatment of patients with HER2 receptor conversion.
Altogether our preclinical model is a useful tool to identify alternative targets for the therapy of HER2-negative secondary lesions.
If you want more information about our model and work, please click here https://www.nature.com/articles/s41389-021-00360-9?proof=t%25C2%25A0 , read our paper “Evolution of HER2-positive mammary carcinoma: HER2 loss reveals claudin-low traits in cancer progression” and send us your comments and suggestions.
The authors thank M.S. Semprini and S. Roncucci for the precious help in the realization of the "graphic story".





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in