A 22 year old man, with no relevant past medical history or family history, is diagnosed with late-stage colorectal cancer (CRC). He is found to have no genetic predispositions or any traditional risk factors, and he dies three months later.
We are seeing more of these tragic cases, whereby people aged less than 50 years are being diagnosed with CRC - a disease traditionally seen in older people. Incidence of CRC among people aged 20-29 years has been observed to increase annually by 8% across Europe,1 and in the USA, CRC is the third leading cause of cancer death in adult males less than 50 years old.2
Why is this?
We can hypothesise several non-genetic factors, which correlate with this trend, which may be responsible: Increased consumption of ultra-processed foods and drinks high in sugars, fats and chemicals such as monosodium glutamate and titanium dioxide; obesity; sedentary lifestyles; alcohol consumption; and antibiotic usage.3 All have a detrimental impact on the population of microbes living within our guts – known as the gut microbiome. Our gut microbiome contains more microbes than we have cells in our body, and its vital numerous digestive and immune functioning roles has led to its recognition as a “virtual organ” of the human body, playing a key part in maintaining health and preventing disease.4
Taking antibiotics is likely to have a detrimental impact on the gut microbiota.4 Although a crucial treatment which revolutionised public health in the 20th century, unnecessary use can be dangerous. Antibiotics can unintentionally induce gut dysbiosis, which means permanent changes to the natural diversity of bacteria within the gut. This can potentially lead to altered bacterial activity and colonisation of harmful bacterial strains, which have implications on the normal immune-surveillance role of the microbiome.5 Dysbiosis has been associated with obesity and autoimmune diseases, and can lead to chronic inflammation – theoretically increasing risk of cancer.6
Antibiotics are widely used; 69% of infants aged less than two years are exposed to antibiotic therapy in the USA,7 and accessibility is increasing across low and middle-income countries, resulting in frequent unnecessary usage for common childhood infections.8
Dr Leslie Samuel, consultant GI oncologist in Aberdeen, noticed more young, fit, non-obese individuals being diagnosed with CRC and wondered why. Could widespread antibiotic usage be a contributing reason?
I am a final year medical student, with an intercalated Master’s degree in Public Health and a keen interest in cancer epidemiology. I did some previous research with Dr Samuel concerning genetic predisposition and survival among early-onset CRC patients,9 and the subject fascinated me. Working closely alongside a primary care professor at the University of Aberdeen, and statisticians, including Dr Ron McDowell, and a pharmacy professor at Queen’s University Belfast, we devised a case control study using a national primary care database to help answer this question.
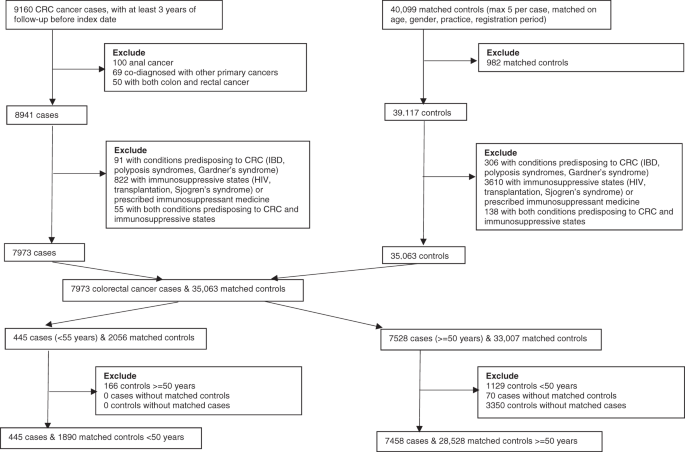
To perform this analysis, we identified 7,903 CRC cases (445 were aged less than 50 years, and therefore termed early-onset) and 30,418 controls. We conducted analysis separately for early-onset and later-onset CRC cases; evidence suggests that risk factors may differ between early-onset and later-onset disease. At the time, only a small number of studies investigating an antibiotic-CRC link existed; these studies had limited their population to older adults and showed mixed results. Our study therefore aimed to determine associations with early-onset CRC, and compare it with later-onset CRC.
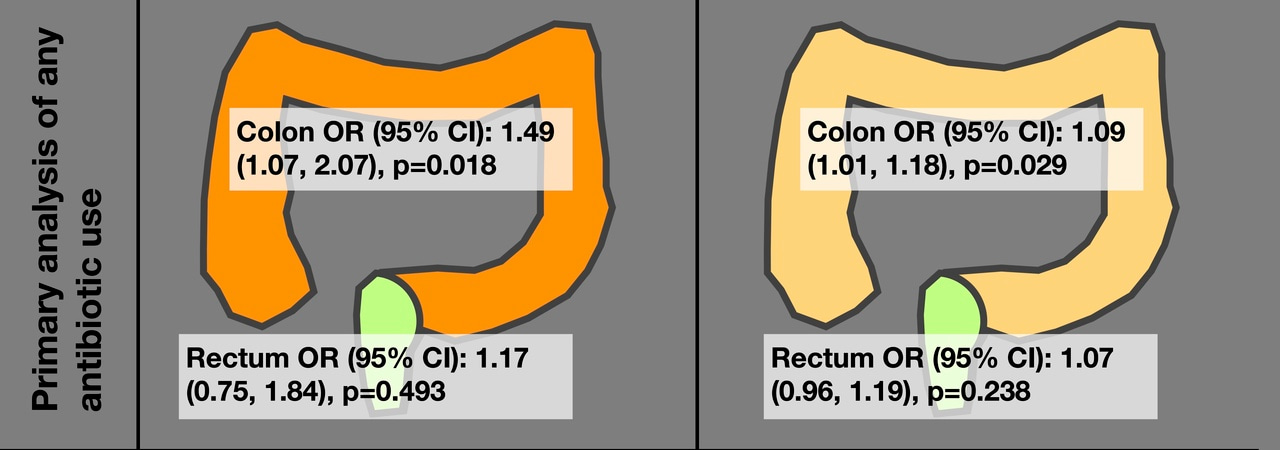
Analysing colon and rectal cancer separately, due to differing risk factors and disease development,10 we found antibiotic consumption was significantly associated with colon cancer among those aged less than 50 years, with an increased odds of 49% (Figure 1). Associations were also significant in people aged over 50 years, who had increased odds of about 9%. We did not find any significant association between antibiotic exposure and rectal cancer. A further subgroup analysis, where we looked instead at proximal and distal colon cancer (i.e. right and left) instead, suggested antibiotic use may be more strongly associated with proximal colon cancer in younger individuals. This may be because the proximal bowel has greater bacterial diversity than the distal bowel, and the microbiome is likely to be more sensitive to antibiotic-induced dysbiosis.11

Figure 1: Associations Results from our primary analyses, showing associations between any antibiotic usage and CRC.
Although we managed to adjust our results for many co-morbidities and other factors which may influence such a relationship, such as diabetes mellitus, body mass index and alcohol consumption, we unfortunately could not adjust for dietary factors as this was not available from the primary care database we used. Another major limitation of our study was sample size; although CRC in people aged less than 50 years is more common than it used to be, it is still relatively rare which impacted on the power of our results.
In conclusion, we found antibiotic exposure was associated with colon cancer among all age groups. This, along with multiple other dietary and lifestyle factors, may be contributing to increased cases of colon, but not rectal, cancer among young people. Further studies are required to better understand the role of the gut microbiome in the development of colon cancer. Regardless of our findings, antibiotic prescribing should be judicious due the pressing concern of antimicrobial resistance. Healthcare professionals and members of the public need to be aware that unnecessary antibiotic usage, especially among young people, should be avoided.
Personally, I am excited to see further work evaluating the role of the gut microbiome in the development of colon cancer and what we can do to modulate it. Probiotic supplements, which vary in preparation, contain strains of live microbes and aim to improve or restore the gut microbiome. Could pairing an antibiotic with a probiotic drug limit the incidence of gut microbiome dysbiosis, given that antibiotic exposure is often necessary and unavoidable?
References:
- Vuik, F. E. R. et al. Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut 68, 1820–1826 (2019).
- Siegel, R. L. et al. Colorectal cancer statistics, 2020. CA. Cancer J. Clin. 70, 145–164 (2020).
- Hofseth, L. J. et al. Early-onset colorectal cancer : initial clues and current views. Nat. Rev. Gastroenterol. Hepatol. 17, (2020).
- Zitvogel, L., Daillère, R., Roberti, M. P., Routy, B. & Kroemer, G. Anticancer effects of the microbiome and its products. Nat. Publ. Gr. 15, 465–478 (2017).
- Whisner, C. M. & Aktipis, C. A. The Role of the Microbiome in Cancer Initiation and Progression : How Microbes and Cancer Cells Utilize Excess Energy and Promote One Another’s Growth. Curr. Nutr. Rep. 8, 42–51 (2019).
- Kasselman, L. J., Vernice, N. A., DeLeon, J. & Reiss, A. B. The gut microbiome and elevated cardiovascular risk in obesity and autoimmunity. Atherosclerosis 271, 203–213 (2018).
- Bailey, L. C. et al. Association of antibiotics in infancy with early childhood obesity. JAMA Pediatr. 168, 1063–1069 (2014).
- Allwell-Brown, G. et al. Trends in reported antibiotic use among children under 5 years of age with fever, diarrhoea, or cough with fast or difficult breathing across low-income and middle-income countries in 2005–17: a systematic analysis of 132 national surveys from 73 countrie. Lancet Glob. Heal. 8, e799–e807 (2020).
- Perrott, S. et al. Young-onset colorectal cancer in the North East of Scotland: survival, clinico-pathological features and genetics. BMC Cancer 20, 108 (2020).
- Cisyk, A. L., Nugent, Z., Wightman, R. H., Singh, H. & McManus, K. J. Characterizing Microsatellite Instability and Chromosome Instability in Interval Colorectal Cancers. Neoplasia 20, 943–950 (2018).
- Dejea, C. M. et al. Microbiota organization is a distinct feature of proximal colorectal cancers. Proc. Natl. Acad. Sci. U. S. A. 111, 18321–18326 (2014).




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in