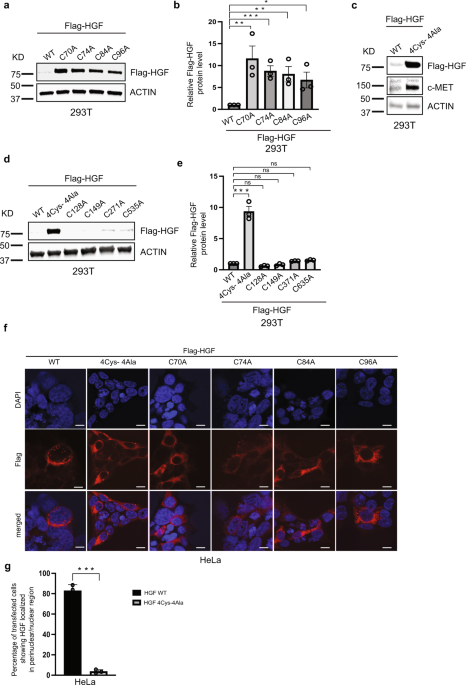
Immunosuppression is an important biological phenomenon that regulates both physiological and pathological processes in cells. To delineate the mechanism better, we sought to identify a reliable and unifying “biomarker” associated with the proteins involved in immunosuppression. To achieve that goal, we scanned the protein databases, InterPro (https://www.ebi.ac.uk/interpro/) and UniProt (https://G-LecRK.uniprot.org), and found > 28,000 proteins across eukaryotes, archaea, bacteria and viruses carrying a unique PAN domain. Interestingly, our GO-enrichment analysis confirmed that many of these identified proteins are involved in diverse immune signaling processes and bear striking similarities with cysteine-rich peptides (CRPs) that have already been widely implicated in regulating immune responses as this domain contains 4-6 cysteine residues. Here we show that these four cysteine residues in the PAN domain are essential for the activation HGF/c-MET signaling cascade as it provides the binding site for the c-MET and regulates the internalization and ubiquitination of c-MET in cells. Our biotinylation cross-linking experiment confirmed a direct link between the PAN domain and the c-MET interaction. Mutations in each of the four cysteine residues equally impaired the signaling cascade, whereas altering any cysteine outside the PAN domain didn’t significantly impact the overall abundance of the ligand, HGF, which was possibly caused due to the inability of its binding with the receptor, c-MET. Moreover, targeting the PAN domain of HGF downregulates the activation of STAT3, a well-known cancer biomarker, as its hyperactivation has been reported in several cancers. Mutations in the core cysteine residues in the PAN domain also alter the pattern of signature gene expression, including genes involved in cancer metastatic and cell cycles, without having any significant impact in the protein structure. Overall, the cancer cell proliferation was reduced by targeting the PAN domain. Given the importance of the HGF pathway in multiple cancers, including glioblastoma, bladder, breast, colon, leukemia, lung, and melanoma, this discovery could open a new avenue in developing targeted cancer drug therapies with limited off-target effects. We also believe our findings will provide opportunities to improve existing treatment targeting the HGF/c-MET cancer axis.
See the full paper in Communications Biology here:

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in